|
In this project we were being tasked with creating a water filter out of simple materials, in order to understand the chemistry behind cleaning waste water. We started our project by coming up with a brainstorm of all the different ways that we could clean water, and from there we picked the best one that our group could build in the time allotted. Next, we had to discuss the ideas of how our product could actually help people or have an impact. This is where we came up the problem of people in underdeveloped countries not having access to drinking water, which would be the target audience for our filter. Keeping this in mind we did our best to construct a prototype
|
of our design that could be built affordably and also be accessible to these people. We continued on with our process by collecting materials to build the filter, and after it was built we put dirty water through it. One of the main issues with our project was finding water that was dirty enough to test. We spent a lot of time using water for tests that turned out to not be reliable, so we had to start over. This caused some issues with our time management and we had to work a lot more efficiently to finish our presentation and data collection on time. Next, after and before running the water through the filter we took tests to see what the filter changed about the contents of the water. We recorded these results to conclude our findings in our final step. The last step was communicating our results/findings by presenting to a panel of judges from our local water treatment facility in Novato. The slideshow we presented can be located below.
Driving Questions
Below are some of the driving questions that my group and I came up with in the early planning stages of our project. These helped direct our efforts and research.
Below are some of the driving questions that my group and I came up with in the early planning stages of our project. These helped direct our efforts and research.
- What kind of filters are we going to use?
- What kind of chemicals will we use?
- What would the price look like for this item?
- How will it be accessible to people in developing countries?
- How will it be sustainable or environmentally friendly?
- What kind of table display will convey our message or problem?
- How large should the item be?
- How much will it weigh?
- What things that are similar to this currently exist?
Our Problem
We hoped to help third world countries successfully gain access to clean drinking water that is affordable. With our model we wanted to provide people without clean water with an easy solution so they can stay healthy and live prosperous lives. Of course this is a part of the hypothetical future stages of our plan, but we kept it in mind to meet the goal. Some of the facts below were what we found during our research about our topic:
These facts and numbers really put a face to our problem and made us even more motivated to come up with a functioning and reasonable solution to possible aid these people in need. The logic we had was that if we are fortunate enough to have access to clean water, we should focus our efforts into making sure every person has the same luxury.
We hoped to help third world countries successfully gain access to clean drinking water that is affordable. With our model we wanted to provide people without clean water with an easy solution so they can stay healthy and live prosperous lives. Of course this is a part of the hypothetical future stages of our plan, but we kept it in mind to meet the goal. Some of the facts below were what we found during our research about our topic:
- People in developing or poverty stricken countries do not have complete access to clean water
- 785 million people lack even a basic drinking-water service, including 144 million people who are dependent on surface water.
- In least developed countries, 22% of health care facilities have no water service, 21% no sanitation service, and 22% no waste management service.
- Drinking unclean water can lead to serious diseases and even death
These facts and numbers really put a face to our problem and made us even more motivated to come up with a functioning and reasonable solution to possible aid these people in need. The logic we had was that if we are fortunate enough to have access to clean water, we should focus our efforts into making sure every person has the same luxury.
|
Planning
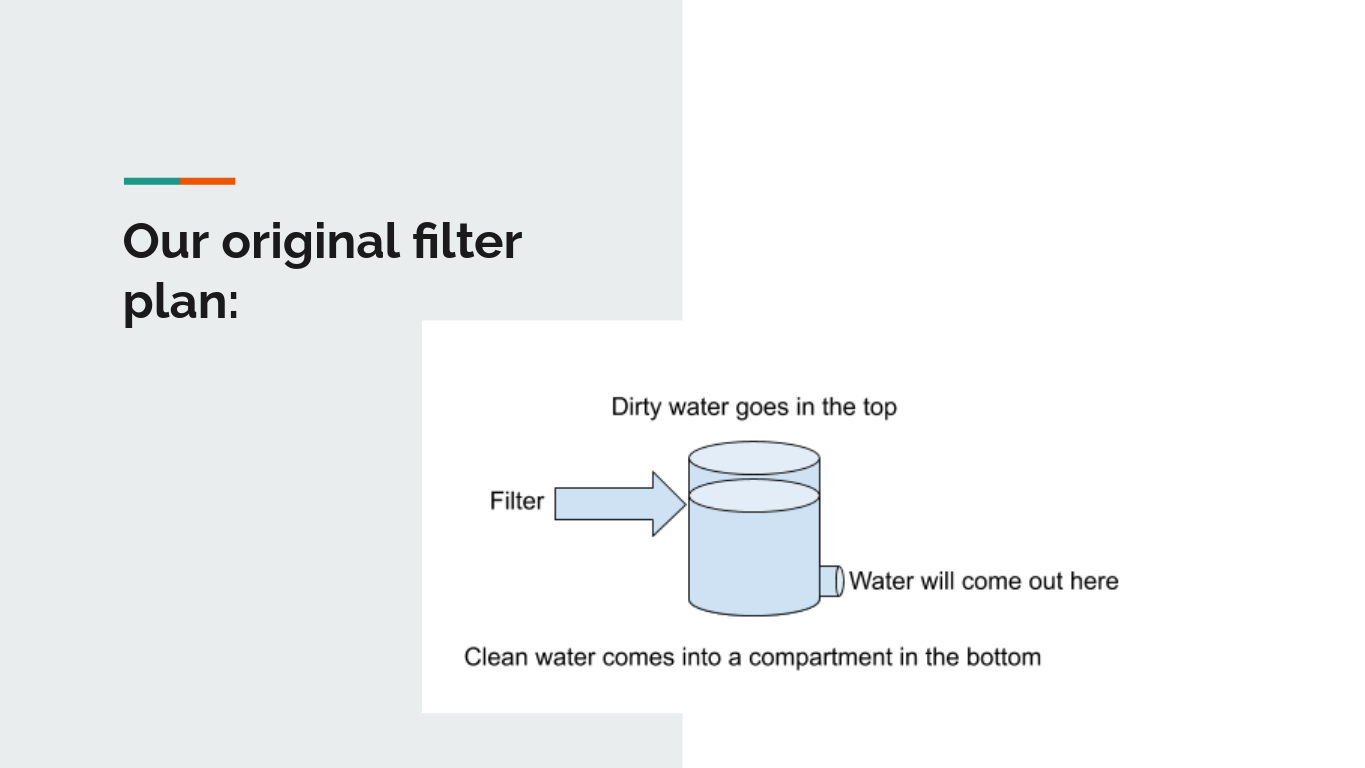
To the right is an image of our construction plan for the filter we constructed. It has three layers, and is made out of some repurposed cups and containers. The three layers consisted of activated cotton balls, charcoal, sand, and gravel (bottom to top). The materials we used were a green plastic cup that we chiseled the bottom and top off of so it was open for liquid to be poured through, and a 2 quart container where the water was collected. The lid of the 2 quart container had multiple holes that the filtered water could flow through, and also held some filter paper for extra reinforcement of fine particles getting into the water. |
An image of our original plan
|
Our Materials
Background
For background we first started by researching the water crisis in Flint, Michigan. The city was in the process of building a new pipeline system to bring water from Lake Huron, a different location or water source. The water almost immediately became discolored and foul smelling. What caused the crisis?
For background we first started by researching the water crisis in Flint, Michigan. The city was in the process of building a new pipeline system to bring water from Lake Huron, a different location or water source. The water almost immediately became discolored and foul smelling. What caused the crisis?
|
Trihalomethanes
|
Lead
|
To get into more detail about the membrane, it is essentially a protective layer that separates the pipe material from the water flowing through. It was important to learn about this information, as it showed us how chemistry can affect the water we drink. We took this into account with our filter we built to try and make the water we put through it as clean and drinkable as possible.
To Better Understand
Solution: a liquid mixture that is dissolved. The dirty water we used in our filter would be the solution.
Solute: a component in a solution that is dissolved in the solvent. The solutes in this project would be all the particles in the dirty water we were trying to get rid of.
Solvent: able to dissolve other substances. Water is known as a universal solvent, and this is what we used in our experiments and put through our filter.
Ionic vs Molecular: an ion is an atom or group of atoms that has a positive or negative charge (it has different number of electrons). An ionic bond is the transfer of electrons between a positive and negative (metal and nonmetal). Molecular bonds are between a metal and another metal that involves the sharing of electron pairs between atoms.
Polarity: A covalent bond in which electrons are not shared equally with a negatively charged and positively charged end. Water is extremely polar (oxygen and hydrogen)
Solubility: the ability of a substance to dissolve something
Wastewater: this is a term used to describe dirty or contaminated water that is not used for drinking or other things. This was part of our project, as this is what we essentially tested our filter with.
Solution: a liquid mixture that is dissolved. The dirty water we used in our filter would be the solution.
Solute: a component in a solution that is dissolved in the solvent. The solutes in this project would be all the particles in the dirty water we were trying to get rid of.
Solvent: able to dissolve other substances. Water is known as a universal solvent, and this is what we used in our experiments and put through our filter.
Ionic vs Molecular: an ion is an atom or group of atoms that has a positive or negative charge (it has different number of electrons). An ionic bond is the transfer of electrons between a positive and negative (metal and nonmetal). Molecular bonds are between a metal and another metal that involves the sharing of electron pairs between atoms.
Polarity: A covalent bond in which electrons are not shared equally with a negatively charged and positively charged end. Water is extremely polar (oxygen and hydrogen)
Solubility: the ability of a substance to dissolve something
Wastewater: this is a term used to describe dirty or contaminated water that is not used for drinking or other things. This was part of our project, as this is what we essentially tested our filter with.
Water filter is a general term that refers to any system or process that is used to filter out particles and pollutants from water. Also anything that removes any amount of particles, sediment, bacteria, and the chlorine taste is called water filtration. Our focus was on turning waste water in useable and drinkable water, so we looked at what a professional and large scale filtration system would look like. What happens a water filtration plant is that contaminated water passes through screens made of mesh or other polymers to remove large particles such as sticks, leaves, and other debris. Next it moves through a series of filters consisting of sand and sometimes even biological filters such as algae to remove smaller contaminants, such as dangerous bacteria. The next part is disinfection which includes adding a small amount of powder or liquid containing chlorine to help kill pathogens or bacteria. Then it has a stage where it moves the water through ultraviolet light which rapidly kills any remaining harmful pathogens that may have passed through the filters. The finished product is clean water.
|
We modeled out filter off of this idea essentially. We chose the layers of our filter extremely carefully. The gravel was for getting rid of large items/dirt, and the sand was for filtering out smaller particles. The third layer was activated charcoal, which did most of the heavy lifting. Activated charcoal is created by changing the internal structure of charcoal through high temperatures. This change in structure comes in the form of smaller pores. It has a negative electrical charge which helps it attract positively charged molecules such as toxins that can be
|
Image of our filter we constructed showing the different layers
|
found in water. Through this attraction these toxins are absorbed by the charcoal. It is still safe to consume this charcoal because it is not absorbed by our bodies so it will carry the absorbed toxins in and out of our digestive tract without any harm. Activated charcoal is not good at removing bacteria or viruses, which is why we concede to the fact pur filter can still improve (this was be discussed later). Also, many organic impurities in water are carbon based and attracted to the carbon in the charcoal creating chemical bonds between the two. Other compounds, such as water, do not bond to the charcoal so they are able to pass through.
Our Tests
|
To the left is an image of our water sample that we used and put through our filter. Our goal was to make the water more clear, and ultimately safe to drink. As mentioned earlier it took us a few trials before we were able to find a water sample that was dirty enough to put through our filtration system. However, after searching our campus, we were able to find some water that was contaminated enough to be tested. |
We ran several different on tests on our water sample before filtration, and again after so we could compare the results. The tests that we took include an ammonia nitrogen test, pH levels, an iron test, a copper test, and a chlorine test.
Our Data
Ammoniacal nitrogen is a measure for the amount of ammonia, a toxic pollutant often found in landfill leachate and in waste products, such as sewage, liquid manure and other liquid organic waste products. The Ammonia Nitrogen levels stayed the same for the dirty sample before and after filtering at around 0.1 ppm which is within the safe levels. Ammonia is dangerous to aquatic life but it is fine in human drinking water. The chemical composition of this compound is NH3.
pH is defined as the potential of hydrogen, or how much hydrogen is in a substance. It can be measured on a logarithmic scale that ranks the relative basicity or acidity of substances to other substances. The basicity and acidity is based off of the hydrogen ion activity in the substance.
pH is defined as the potential of hydrogen, or how much hydrogen is in a substance. It can be measured on a logarithmic scale that ranks the relative basicity or acidity of substances to other substances. The basicity and acidity is based off of the hydrogen ion activity in the substance.
- High pH = low hydrogen ion concentration (basic)
- Low pH= high hydrogen ion concentration (acidic)
|
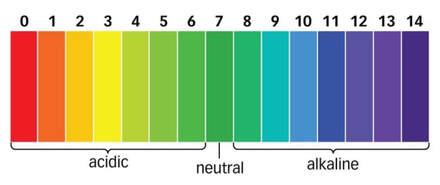
To the left is a simple image of what a pH scale looks like. It is good to note that the pH of "pure" water is 7. The average pH range for safe drinking water is somewhere between 6 - 8.5. If the substance is too acidic, that causes health problems and the water is unsafe to drink. The same idea applies to high pH levels, which are also not good for you to drink if they are too high.
|
- pH of dirty water: 7
- pH of water after filtration: 8
While the pH of the water after filtration increased, this still falls under the range of safe levels in drinking water. Additionally, while you do not want super basic drinking water, it is a good sign that the pH is not dropping to become more acidic which is very harmful and unsafe to drink.
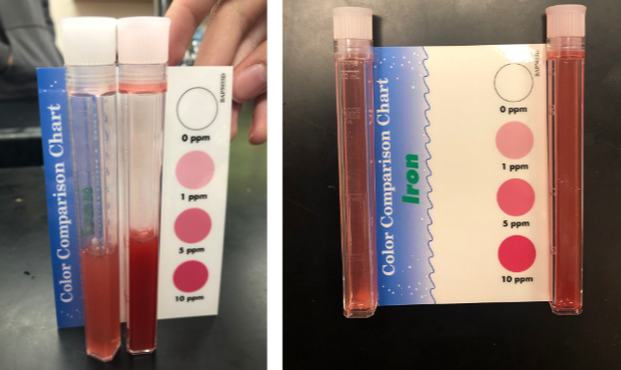
Iron occurs naturally in nature and can very easily come into water whether it be from natural pools or drinking water. The presence of iron can affect the taste of water, and may also alter the appearance or how clear the water could be. Testing for iron is good for monitoring water, and it also checks for waste materials. Iron in drinking water contains bacteria that feeds off of it and high concentrations of iron can lead to overload which causes diabetes, stomach problems, vomiting and nausea. The safe level of iron in drinking water is 0.3 parts per million (ppm).
This was a very exciting result to get back! This showed us that our filter did work in a sense, as it lowered the high amount of iron by half of its original value. While this is still not in the safe level of iron (0.3 ppm) this test was done after only one pass through the filter. Therefore we can predict that the presence of iron will continue to decrease the more times we put it through. Luckily, the way our filter is designed we can take that filtered water and run it through the whole process multiple times. |
The filtered sample is on the left and dirty is on the right
|
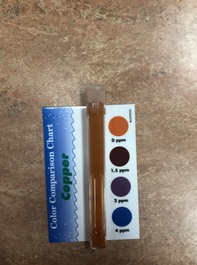
Copper is a chemical element with the symbol Cu and atomic number 29. A high concentration of copper can lead to vomiting, diarrhea, stomach cramps, nausea, liver damage and kidney disease. This is not something that we would want to have a lot of in our water we drink, so this was another important test we wanted to conduct. The safe level of copper in water is about 1.3 ppm.
Our water sample before filtration had no copper in it, and the filtered water did not have any either. We cannot definitely say how our filter deals with copper, since the sample had none to begin with. Expect it is promising that none was added or came out in the filtered sample. |
Chlorine is one of the lightest halogens on the periodic table and is abbreviated at the symbol Cl. A high concentration of chlorine has been linked to higher incidence of bladder, rectal and breast cancer. As a halogen, chlorine is a highly efficient disinfectant, and is added to public water supplies to kill disease-causing pathogens, such as bacteria, viruses, and protozoans, that commonly grow in water supply reservoirs, on the walls of water mains and in storage tanks. This however is not something that you would want to drink too much of in your water and put into your body.
- Dirty water: 0 ppm
- Filtered water: 0 ppm
Further Steps
Obviously, our filter still needs some modifications to be at the standard that we want it to be at. Some of the next steps for our filter and additions we want to make are to make sure that if this was to be a product that people used it would be reliable and efficient.
Reaction equation: NH3 + HOCI -> NH2Cl + H2O
During this reaction three forms of chloramines form. When the pH is 7 or above monochloramines form. This is more beneficial than chlorine because it lasts much longer than chlorine.
- Replace filter paper (it is not very strong)
- Check for bacteria
- Polish up appearance
- Larger Scale
- More tests
Reaction equation: NH3 + HOCI -> NH2Cl + H2O
During this reaction three forms of chloramines form. When the pH is 7 or above monochloramines form. This is more beneficial than chlorine because it lasts much longer than chlorine.
Water Table
Another part of our assignment was creating an interactive water table that kids could come over to at a science fair and participate in an experiment. For our display observers would be allowed to create "dirty" water themselves. They will then pour their concoction through our filter to observe how it is able to produce clean drinking water. I say "dirty" water, because we aren't actually going to be putting dirt into the water. Instead we decided to have the participants squeeze lemons into a glass of water, and have the pulp and seeds act as the big dirt they could find in contaminated water. Then they will pour it through the filter and see how all the big chunks get filtered out, and the product is cleaner water. It may have a bit of a citrus taste to it, but it gets the basic idea of our filter without putting anyone in harm if something goes wrong.
Reflection
Two things that I did well in the project was time management and collaboration. The first skill that I performed well was time management. A specific example of when this was evident was when our teacher informed us at the beginning of our project that we did not have a lot of time to complete our filter. This skill came in handy when we had to come up with a construction plan for building and allowed us to organize when we would be conducting our testing. The second skill that I utilized in this project was collaboration. A specific example of me using this skill well was towards the end of our working days. I wanted to make sure that my group and I finished everything on time, so we talked out the situation and assigned tasks to people to work more efficiently. I worked on the presentation we would be giving, by creating slides and compiling the data that some of my other teammates had collected during our experiments. We were really working like a well oiled machine, and this helped us finish the project in a timely matter and efficiently.
Two skills that I could still improve upon would be communication and decision making. The first skill that I could still improve upon is communication. A specific example of when this area became a problem was the day of our presentation. My group and I had been split up during tutorial, and this was the time where we were going to finalize the presentation we had made and practice. We were unable to do so, and trying to communicate all of the logistics from different parts of the school made this a big challenge, especially right before we presented. In the future I will make sure that my group and I work together to make a solid time to practice our presentations and a better form of communicating. The second skill that I still need to work on is decision making. I would definitely say that I am an indecisive person, and this was highlighted in this project. A specific time when this caused a problem was at the beginning of our project during brainstorming. We had to pick a topic to focus on for our problem and water filter design, but my group and I could not pick one thing to do. It was frustrating, because we all could not make a decision which made the process lengthy. In the future I will be careful with my decisions, but also be more confident in what my gut is telling me to go with.
All in all, this was a super fun project! I learned a lot about water treatment, and how much more complicated it is for water to be cleaned than I originally thought. Most importantly it taught me to be grateful for what I have in life, like clean water, which some people are not as fortunate to have. I loved being able to focus our project to people in need, and hopefully in the future these kinds of technology and clean water will be universally available.
Two things that I did well in the project was time management and collaboration. The first skill that I performed well was time management. A specific example of when this was evident was when our teacher informed us at the beginning of our project that we did not have a lot of time to complete our filter. This skill came in handy when we had to come up with a construction plan for building and allowed us to organize when we would be conducting our testing. The second skill that I utilized in this project was collaboration. A specific example of me using this skill well was towards the end of our working days. I wanted to make sure that my group and I finished everything on time, so we talked out the situation and assigned tasks to people to work more efficiently. I worked on the presentation we would be giving, by creating slides and compiling the data that some of my other teammates had collected during our experiments. We were really working like a well oiled machine, and this helped us finish the project in a timely matter and efficiently.
Two skills that I could still improve upon would be communication and decision making. The first skill that I could still improve upon is communication. A specific example of when this area became a problem was the day of our presentation. My group and I had been split up during tutorial, and this was the time where we were going to finalize the presentation we had made and practice. We were unable to do so, and trying to communicate all of the logistics from different parts of the school made this a big challenge, especially right before we presented. In the future I will make sure that my group and I work together to make a solid time to practice our presentations and a better form of communicating. The second skill that I still need to work on is decision making. I would definitely say that I am an indecisive person, and this was highlighted in this project. A specific time when this caused a problem was at the beginning of our project during brainstorming. We had to pick a topic to focus on for our problem and water filter design, but my group and I could not pick one thing to do. It was frustrating, because we all could not make a decision which made the process lengthy. In the future I will be careful with my decisions, but also be more confident in what my gut is telling me to go with.
All in all, this was a super fun project! I learned a lot about water treatment, and how much more complicated it is for water to be cleaned than I originally thought. Most importantly it taught me to be grateful for what I have in life, like clean water, which some people are not as fortunate to have. I loved being able to focus our project to people in need, and hopefully in the future these kinds of technology and clean water will be universally available.